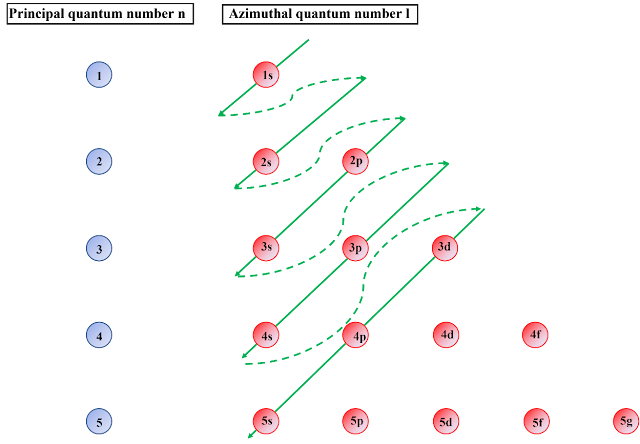
Electrons configuration in Atom as per Quantum Mechanics
To understand the movement of electrons in a semiconductor device, we need to understand the electronic configuration of an atom and how it interacts with the crystal lattice. We already know that atom consists of Neutrons (neutral charge) and Protons (positive charge) inside a nucleus & Electrons (negative charge) revolved around the nucleus (Figure 1).

Figure 1 Atomic structure
Note: (Just for your information) If you remember in the intermediate level or BTech 1st year, we have studied in quantum mechanics that these electrons are restricted to certain energy levels (quantized). In this regard, Plank’s blackbody radiation and Einstein’s photoelectric effect established the discrete nature of light (quantization). In the late 1920s, quantum mechanics had developed in which Heisenberg developed matrix mechanics and Schrödinger developed wave mechanics at the same time. Although the mathematical formulations are quite different, the basic principles are identical.
Although The Schrödinger equation describes the interaction between particles (e.g. electrons) and potential field, it’s enormously difficult to solve the Schrödinger equation in the case of complicated atoms (Atomic number greater than 1 or you can say the total no. of electrons in a neutral atom). Therefore, the solution of the hydrogen atom (atomic number 1) is significant to understand the permissible energy states for electron arrangements.
It is well known that a hydrogen atom consists of a single electron and a proton system. We can call this system a two-particle system. In some circumstances, the two-particle system's motion can be described as the motion of a single particle with respect to a center of mass. The mass of the particle is referred to as a reduced mass with respect to the center of the mass system. In this consideration, we’ll assume that the electron is relatively moved around the proton, and the proton is located at the center (in this case at the origin). Since protons have higher mass with respect to the electron, therefore we will consider the reduced mass is equal to the electron mass, and the proton is located at the origin.

Figure 2 (a) Center of mass concept. (b) Position of an electron with respect to proton (Origin)
Note: - I know you may be thinking what is this. Why are we assuming so many things and nothing is fixed? Basially, in an atom when we say that Proton is in center, then its important to understand which center are we talking about!
Let me explain you in my (Puneet's way) and you try to visualise it. (People or scientist may not like it but at least you can understand it. From analogy point of view you can consider it's a cloud system. When we see a cloud can we say that it's a 1 cloud of this size, this is the boundry and this is center of cloud. but still we see that whole piece of cloud moving together and merging with other piece of cloud and they have center also and outer periphery also. So this ATOM structure is like that. Now, Try to understand.
- A Cloud system having more denser at the center (which is also moving) or say that become center of whole cloud (center of the mass system).
- This cloud portion which is present in the center we are saying it's Proton.
- Rest of the portion of cloud, you can say it's electron. (remember this I am trying to explain you for Hydrogen atom)
- Now, you can say that if these are 2 things, can we separate out but that's not going to happen or I say if it will separate out then whole system collapse, so I (scientist) said that both are related to each other (one as positive and other as negative). if you seperate out these 2, whole cloud system collapse. So, when these 2 are together then we say it's an ATOM.
- But still outer part of Cloud can be separate out from Center part and someother part can come and replace it. So, with respect to center part, outer part (electron) is tied with center part (proton).
Now, Read once again the above one and then below one. You will get more clearity.The electrons and nucleus (especially protons) are interacted with by the Coulombic potential


Figure 3 Spherical polar co-ordinate system
Let’s try to understand a few key points/equations of Quantum mechanics. If you are interested, you can go into detail by referring to the standard textbooks. Here we are just capturing for reference purposes.
Note:- If you want to skip, go to equation no 11 directly but I will recommend to read it once.
Assume that the potential distribution is spherically symmetric, a time-independent three-dimensional Schrödinger equation in spherical coordinates can be written as



Note:- Hold ON - If you are thinking about so many equations - It's okay. All these are important (may be not today but it will be one day). I will explain you. Just read it once.

The negative expression specifies that the electrons are bound with the nucleus and the energy is quantized. It should be noted that the value of n should not be zero as the energy expression become invalid. Therefore, n should be an integer.
Note:- Puneet's Way: -
When Scientist studied it further they come to know the cloud is not in 2D and presence of outer area of cloud has some pattern in 3D (Dimensional). Also they figured out that outer surface have some motion and usually this motion is around the inner surface (neculus system - dense part) and this moment is not in X, Y, Z direction only but it's in angular form also. Remember - Whole Cloud system is moving but we are talking about the relative motion of outer cloud with respect to inner cloud (mass). and that's the reason of "Spherical polar co-ordinate system" come into the picture to correctly captured this whole system.
Among the three quantum numbers, there is another quantization condition called spin (s) that electrons should fulfill. Every electron has its angular momentum or spin and can be expressed as

So, you might be thinking why all this??
I will say, have patience and we will explain all this to you in coming articles. This is an intro article. This will help you to understand the moment of electrons in the other orbits (also in outer most orbit, which is very important to understand as we always talk about Valency electron and Valence Band).
By -
First Author - Dr. Bhaskar Patnayak
Second Author (Editor) - Mr. Puneet Mittal